Six British Biotechs Helping to Tackle COVID-19
Table of contents
The Org looked at British biotech companies that have made leaps across a number of areas, from antibody testing to gene-sequencing, and who have boosted sales and innovation capacity in response to COVID.

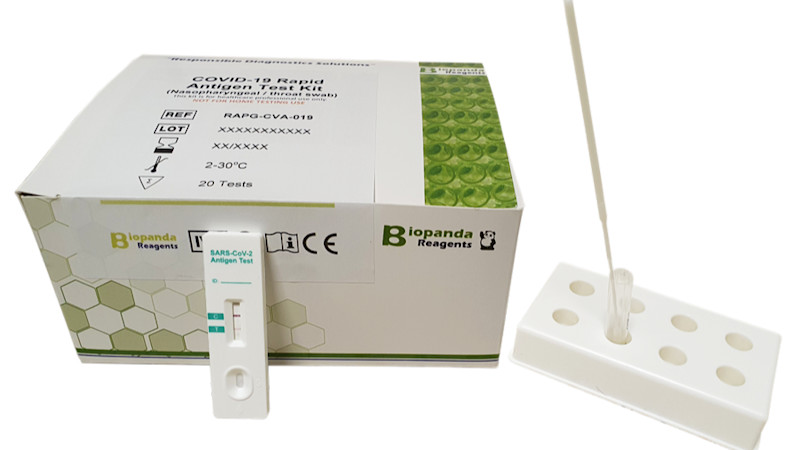
Biopanda Reagents
Belfast-based Biopanda made the news last summer when it became one of the primary suppliers of antibody tests available. By May, it had sold over 300,000 across the world, mostly in the UK, where it was only available for sale to healthcare and clinical staff. The tests worked by taking a small blood sample and detecting the antibodies one’s body would produce if infected with the virus - called IgM, detectable a few days after infection. The company, which saw a huge demand when rolling out the tests, had to hire extra staff just to help with packing and dispatching and was forced to stop taking international orders due to overwhelming demand.
Biopanda has since gone on to develop a rapid antigen test, intended for use as a tool to assist in the diagnosis of the virus.
Cobra Biologics
Bioservices company Cobra Biologics has been instrumental in helping roll out one of the UK’s leading vaccines, AZD1222, developed by the University of Oxford and AstraZeneca. Cobra and other companies formed a consortium that aided the quick and efficient large-scale manufacturing of the vaccine. Oxford University and AstraZeneca didn’t have any large-scale vaccine manufacturing experience, so production partnerships with Cobra Biologics and others in the consortium was vital in rolling it out.
The biotech’s specialisation in viral vector manufacturing has been its leading contribution to the production of the AZD1222 vaccine. Viral vectors are tools which scientists use to deliver genetic materials into cells, which helps in the development of vaccines.
Last month Cobra was shortlisted for two Bionow Awards - a prestigious accolade in the industry - for its work in developing a leading vaccine.
In October of last year, Cobra also began work with Scancell, who have been working on a variant-proof vaccine, which has the potential to fight off all strains of the virus; clinical trials are due to start within weeks.
Mologic
Describing itself as a “leading developer of lateral flow and rapid diagnostic technologies, products and services,” Mologic works with companies, researchers, and clinicians to develop diagnostic tools. Last year it was awarded £1 million as part of the British government’s £46 million Covid research funding package. Over the past year, Mologic has immersed itself in antigen rapid testing technology.
These are quick tests designed to detect antibody-producing antigens and have been used to tell people whether they are infected with the coronavirus within 10 - 15 minutes. Mologic’s tests were also trialed at Heathrow airport amongst other large venues over last summer to help determine the most efficient testing method for when airborne travel is back at full capacity.
This month, the biotech company announced a commercial partnership with biotherapeutic developer Avacta Group to accelerate marketing of the latter’s AffiDX™ SARS-CoV-2 lateral flow rapid antigen test, helping to support its accessibility in low and middle income countries.
Randox
Randox is a diagnostics specialist biotech that has been a key player in the development of PCR (Polymerase chain reaction) Covid tests, which works by detecting the virus’ RNA; its genetic information.
Randox’s rapid testing has been used within the UK’s national testing programme and is responsible for some 17% of all the tests carried out. Last month, the biotech said it reached a key milestone of reporting nine million tests to the national programme, and will soon be closing in on 10 million.
It has employed over 850 new staff into its labs and is now able to run through 100,000 samples in less than 24 hours. Radox has gone from doing 150 samples with 10 people on rota in a day last March to 104,000 samples with 850 people last month.

Oxford Nanopore
While most biotech’s have contributed their impressive research towards testing equipment, Oxford Nanopore has used its DNA sequencing technology during the pandemic to help researchers determine coronavirus-causing gene changes.
This has been useful in investigating the different variants of the virus around the world. Oxford Nanopore does its DNA sequencing through a hand-held device called MinION - it’s no bigger than a cell phone at just over 100 grams, and costs $1,000, cheaper than a lot of lab equipment around the world. It runs by simply plugging into a laptop through a USB cable.
The company’s technology has been used in over 50 countries since January 2020 to track the different strains and mutations of the virus. Oxford Nanopore has attracted international investors as a result, with China Construction Bank and the Government of Singapore Investment Corporation (GIC) as investors.
Last October, International Holdings Company invested £39m in a £84.4m equity raise. Oxford Nanopore now has a valuation of £1.5 billion and is one of the UK’s 18 unicorns.

Oxford Optronix
Oxford Optronix wasted no time in supporting the NHS. After an emergency call by the National Health Service, the research instrumentation provider developed an oxygen monitor prototype after only five days of work last March.
Oxygen monitors are classed as Positive Airway Pressure (cPAP) devices, which the NHS was in dire need of at the beginning stages of the pandemic to help provide patients with a steady flow of oxygen.
By April, it had built 2,000 of the Flo-Ox monitors in only eight days.

--
The Org is a professional community where transparent companies can show off their team to the world. Join your company here to add yourself to the org chart!
In this article


The ORG helps
you hire great
candidates
Free to use – try today



